
Consumer Insights
Uncover trends and behaviors shaping consumer choices today
Procurement Insights
Optimize your sourcing strategy with key market data
Industry Stats
Stay ahead with the latest trends and market analysis.
According to World Cancer Research Fund International, pancreatic cancer is one of the most common cancer globally. The pancreatic cancer treatment options depend on the location and stage of the disease. Major companies and research institutes are working towards developing innovative treatments for pancreatic cancer treatment including development of combination drugs (stereotactic body radiotherapy (SBRT), among others.
Pancreatic Cancer Drug Pipeline report by Expert Market Research gives comprehensive insights on the pancreatic cancer therapeutic drugs currently undergoing clinical trials and covers various aspects related to the details for each of these drugs under development. The report includes the analysis of over 100 pipeline drugs and 50+ companies. Moreover, the analysis based on efficacy and safety measures outcomes published for the trials will be covered including their adverse effects in pancreatic cancer pipeline landscape.
The detailed analysis of each drug, drug class, clinical studies, phase type, drug type, route of administration and ongoing product development activities related to pancreatic cancer are covered. Moreover, pancreatic cancer collaborations and commercial assessments are offered, helping stakeholders to make informed decisions.
Read more about this report - REQUEST FREE SAMPLE COPY IN PDF
Pancreatic cancer is among the most challenging cancer to treat, due to its typical late-stage diagnosis and aggressive nature. Due to the availability of limited effective treatment, there is a high need for the development of innovative therapies to provide effective treatment outcomes. According to Americ an Cancer Society, average lifetime risk of pancreatic cancer is about 1 in 60 in women and 1 in 56 in men. There are two types of pancreatic cancer - pancreatic adenocarcinoma and pancreatic neuroendocrine tumors (nets). The symptoms include jaundice (yellowing of the skin and eyes), unexplained weight loss, digestive issues such as vomiting, nausea among others. The pancreatic report insights cover the pathophysiology and epidemiology of pancreatic cancer, developing treatment, and the progress and future aspects of ongoing clinical trials for treatment of pancreatic cancer in detail.
The treatment options for pancreatic cancer include chemotherapy, targeted therapy and immunotherapy, radiation therapy and others. The chemotherapy treatment includes adjuvant chemotherapy, neoadjuvant chemotherapy, and palliative chemotherapy. Common chemotherapy drugs include gemcitabine, capecitabine, irinotecan and leucovorin among others. The ongoing research on clinical trials aim to identify efficient therapeutic targets, development of novel pancreatic cancer therapeutic drugs which are showing promising for the treatment of pancreatic cancer.
For instance, George Fisher, MD, PhD, Stanford University, collaborated for clinical trial of immunotherapy drug Keytruda (pembrolizumab) for the treatment of pancreatic tumors which have genetic mutation called a mismatch repair defect. This trial has resulted into the FDA approval for the drug. Another ongoing trial at Stanford University is being conducted for 'in-situ vaccines’ which aims to investigate injecting tiny amounts of immune-stimulating agents into solid tumors directly.
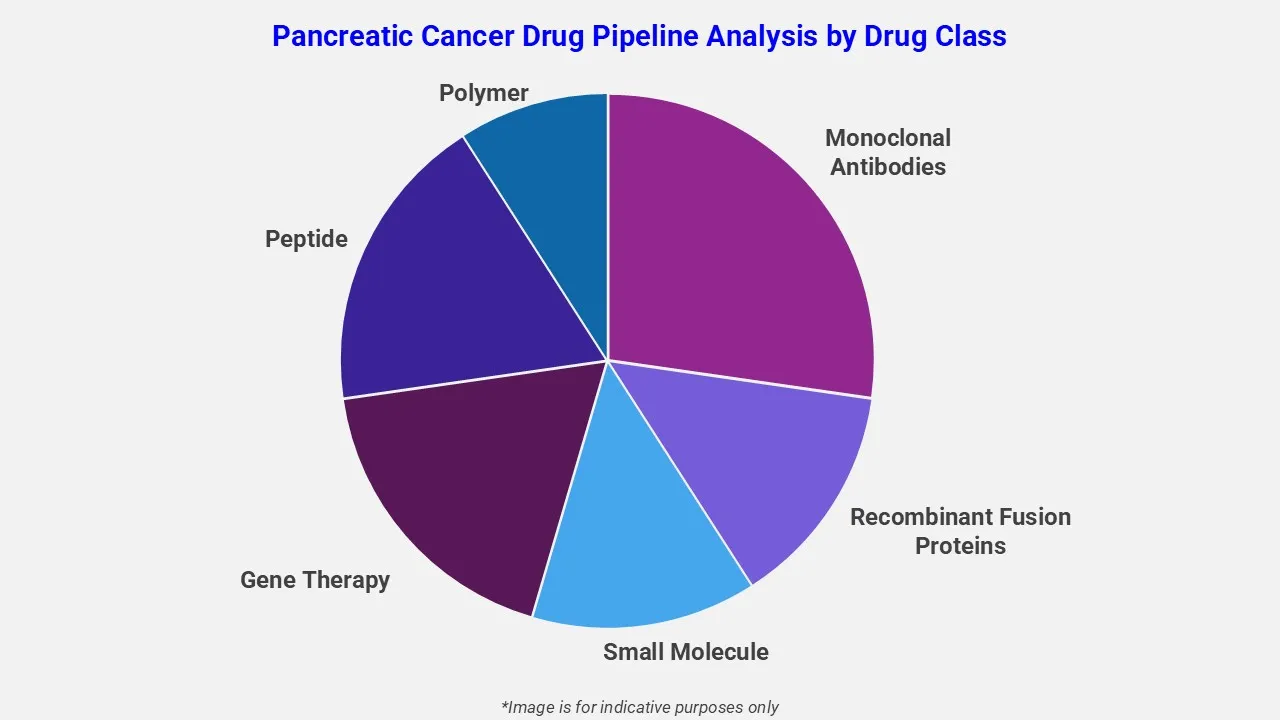
This section of the report covers the analysis of pancreatic cancer drugs candidates based on various segmentations such as:
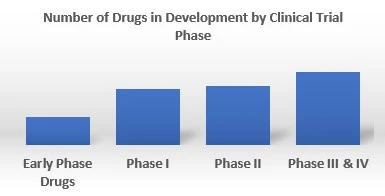
By Phase
EMR’s pipeline assessment report covers 50+ drug analysis based on phase.
By Route of Administration
EMR’s pancreatic cancer therapeutic assessment covers 50+ drug analysis based on route of administration.
The pancreatic cancer pipeline analysis covers phase I, phase II, phase III, phase IV, and early phase drugs for pancreatic cancer. The coverage includes in depth analysis of each drug across these phases. According to EMR analysis, majority of the ongoing clinical trials for pancreatic cancer are currently in Phase II of their development.
University of Texas Southwestern Medical Center is conducting a clinical trial for Pioglitazon e to determine whether addition of the pioglitazone oral medication to standard chemotherapy treatment results in improvement of blood tests.
The route of administration categories covered under pancreatic cancer pipeline report include oral, intravenous, subcutaneous, parenteral and others. The report provides a comparative analysis of the route of administration for each drug in various phases of clinical trials of pancreatic cancer. According to EMR analysis, the oral route accounted for major share of pancreatic cancer clinical trials.
The EMR report for pancreatic cancer pipeline landscape covers the profile of key companies involved in clinical trials and their drugs under development. It provides a detailed pancreatic cancer therapeutic assessment, analyzing the competitive dynamics of the clinical trial landscape. Below is the list of few players involved in pancreatic cancer clinical trials –
This section covers the detailed analysis of each drug under multiple phases including phase I, phase II, phase III, phase IV, and emerging drugs for pancreatic cancer. Moreover, the section covers product description, trial ID, study type, drug class, mode of administration, and recruitment status among others.
Exenatide is currently under observational phase sponsored by AstraZeneca. The study is being done to assess the risk of developing pancreatic cancer who have initiated exenatide among type 2 diabetes mellitus (T2DM) patients.
Sunitinib malate is currently under phase III clinical trials, sponsored by Pfizer. The purpose of the study is to assess the efficiency of sunitinib in patients suffering from advanced pancreatic islet cell tumors. The study will determine the status and size of their tumors, survival, quality of life and safety of the pancreatic cancer therapeutic drug.
Monoclonal Antibody trials is currently under phase I clinical trials, developed by Hoffmann-La Roche. The study is being conducted to assess whether the activation of certain immune system cells will be safe and well tolerated in combination with cytotoxic chemotherapy.
The study is being conducted to identify whether the addition of oxaliplatin to 5-Fluorouracil (5-FU) and Leucovorin (LV) will improve the Progression-Free Survival for pancreatic cancer. This pancreatic cancer drug candidate is currently under phase III clinical trial, being developed by Sanofi.
*Please note that this is only a partial list; the complete list of drugs will be available in the full report.*
Global Cancer Supportive Care Drugs Market
Global Cancer Therapeutics Market
Global Cancer Immunotherapy Market




*While we strive to always give you current and accurate information, the numbers depicted on the website are indicative and may differ from the actual numbers in the main report. At Expert Market Research, we aim to bring you the latest insights and trends in the market. Using our analyses and forecasts, stakeholders can understand the market dynamics, navigate challenges, and capitalize on opportunities to make data-driven strategic decisions.*
Get in touch with us for a customized solution tailored to your unique requirements and save upto 35%!
Explore our key highlights of the report and gain a concise overview of key findings, trends, and actionable insights that will empower your strategic decisions.
| Scope of the Report | Details |
| Drug Pipeline by Clinical Trial Phase |
|
| Route of Administration Oral |
|
| Drug Classes |
|
| Treatment Type |
|
| Leading Sponsors Covered |
|
| Geographies Covered |
|
Mini Report
One User
USD 1,999
USD 1,799
tax inclusive*
Single User License
One User
USD 3,099
USD 2,789
tax inclusive*
Five User License
Five User
USD 4,599
USD 3,909
tax inclusive*
Corporate License
Unlimited Users
USD 5,999
USD 5,099
tax inclusive*
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Small Business Bundle
Growth Bundle
Enterprise Bundle
*Please note that the prices mentioned below are starting prices for each bundle type. Kindly contact our team for further details.*
Flash Bundle
Number of Reports: 3
20%
tax inclusive*
Small Business Bundle
Number of Reports: 5
25%
tax inclusive*
Growth Bundle
Number of Reports: 8
30%
tax inclusive*
Enterprise Bundle
Number of Reports: 10
35%
tax inclusive*
How To Order

Select License Type
Choose the right license for your needs and access rights.

Click on ‘Buy Now’
Add the report to your cart with one click and proceed to register.

Select Mode of Payment
Choose a payment option for a secure checkout. You will be redirected accordingly.
Gain insights to stay ahead and seize opportunities.

Get insights & trends for a competitive edge.

Track prices with detailed trend reports.

Analyse trade data for supply chain insights.

Leverage cost reports for smart savings

Enhance supply chain with partnerships.

Connect For More Information
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
Our expert team of analysts will offer full support and resolve any queries regarding the report, before and after the purchase.
We employ meticulous research methods, blending advanced analytics and expert insights to deliver accurate, actionable industry intelligence, staying ahead of competitors.
Our skilled analysts offer unparalleled competitive advantage with detailed insights on current and emerging markets, ensuring your strategic edge.
We offer an in-depth yet simplified presentation of industry insights and analysis to meet your specific requirements effectively.
Share